News

(Hannover, Germany) Bacteria worldwide keep developing new resistances to antibiotics. Alternative therapies are urgently needed to meet this challenge. To this end, the Fraunhofer Institute for Toxicology and Experimental Medicine ITEM, Leibniz Institute DSMZ-German Collection of Microorganisms and Cell Cultures, Charité – Universitätsmedizin Berlin, and Charité Research Organisation have teamed up and initiated the project “Phage4Cure”. The goal is to establish bacteriophages as an approved drug for treating bacterial infections. The German Federal Ministry of Education and Research is funding this project with almost four million euros over a period of three years.
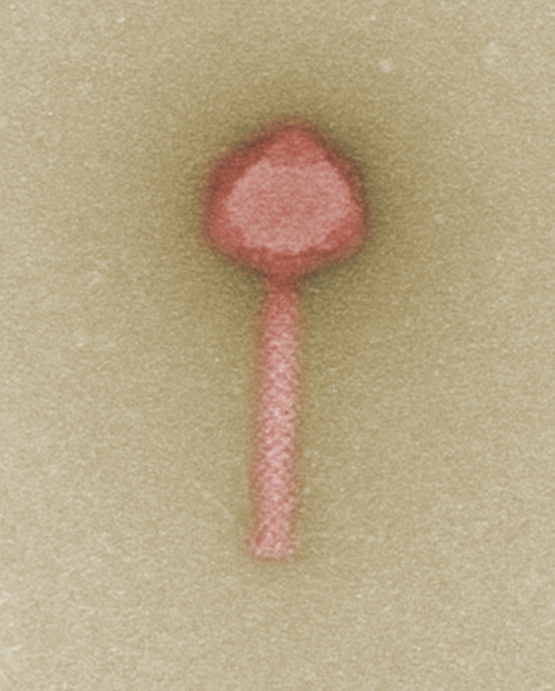 The aim of the German research project “Phage4Cure” is to establish bacteriophages as an approved drug in the fight against infection. Bacteriophages are viruses that highly specifically recognize and bind to a certain type of bacteria, eventually causing their destruction. In eastern Europe in particular, phages have been successfully used for decades already as an alternative or complementary treatment to traditional antibiotic therapy. In the European Union, however, they have not yet been approved as drugs. This is due, among other reasons, to missing quality standards for bacteriophage production, a sine qua non for drug approval by the authorities. Furthermore, systematic clinical trials first have to be performed to demonstrate the safety, tolerability, and efficacy of treatment with phages. This is exactly the aim the project partners are now going for.
The aim of the German research project “Phage4Cure” is to establish bacteriophages as an approved drug in the fight against infection. Bacteriophages are viruses that highly specifically recognize and bind to a certain type of bacteria, eventually causing their destruction. In eastern Europe in particular, phages have been successfully used for decades already as an alternative or complementary treatment to traditional antibiotic therapy. In the European Union, however, they have not yet been approved as drugs. This is due, among other reasons, to missing quality standards for bacteriophage production, a sine qua non for drug approval by the authorities. Furthermore, systematic clinical trials first have to be performed to demonstrate the safety, tolerability, and efficacy of treatment with phages. This is exactly the aim the project partners are now going for.
The scientists will explore bacteriophages specifically targeting the bacteriumPseudomonas aeruginosa. This bacterium frequently becomes multiresistant and can cause, for example, lung infections. “Our goal in the medium term is to develop phages as a medicinal product that can be delivered via different routes of administration, to provide a novel and additional therapeutic option for different infectious diseases – in particular for those cases where antibiotics have reached their limits,” says Dr. Holger Ziehr, project coordinator and director of the Fraunhofer ITEM Division of Pharmaceutical Biotechnology.
In the project “Phage4Cure”, the four partners with their special know-how are each working on a different aspect of this topic. The Leibniz Institute DSMZ working group under Dr. Christine Rohde will identify bacteriophages targeting Pseudomonas aeruginosa, including genetic characterization. “There are many different Pseudomonas aeruginosa strains, with only minor differences between them. The challenge is to identify phages with as broad a spectrum of hosts as possible,” the scientist explains. The phages will then be passed on to Fraunhofer ITEM for further high-quality purification and pharmacological production. The Fraunhofer team under Dr. Holger Ziehr will develop a so-called platform manufacturing process for phage-based therapeutic agents. This means that the manufacturing process can subsequently be used for other phages as well. In addition, the Fraunhofer ITEM scientists will perform preclinical testing.
Further preclinical tests will be conducted at Charité – Universitätsmedizin Berlin by scientists from the team of Professor Martin Witzenrath, Deputy Director of the Medical Department, Division of Infectiology and Pneumonology. Professor Witzenrath is also involved in the design, planning, and performance of the required clinical trials. “Lung infections with antibiotic-resistant bacteria are a clinical problem of increasing relevance. We hope to be able to help patients by means of phage-based therapies in the future,” says Witzenrath. The clinical trial will be conducted in the research unit of Charité Research Organisation GmbH (CRO). Furthermore, CRO will provide organizational and regulatory assistance for the entire project, will keep close contact with supervisory drug agencies, and will take care of data management, statistics, and preparation of the clinical study report. Dr. Andreas Hüser, senior project manager: “Charité Research Organisation has specialized in such studies and we are were excited to be part of this important project. The combination of partners in this project is truly outstanding.”
Short profiles of the project partners and further scientific contacts:
Leibniz Institute DSMZ – German Collection of Microorganisms and Cell Cultures
With over 50,000 cultures of a broad range of microorganisms and cell lines, Leibniz Institute DSMZ is one of the largest bioresource centers worldwide. Its newly founded Department of Bioeconomics and Health is committed to enhancing the phage collection and is working on a variety of research projects on phages.
Dr. Christine Rohde
Bioeconomics and Health, Working Group on Phages
Phone +49 531 2616-220
chr@dsmz.de
Fraunhofer ITEM
Fraunhofer ITEM has more than 25 years of experience in bioprocess development and in GMP manufacturing of biopharmaceutical active ingredients (APIs) and investigational medicinal products (IMPs) for clinical trials.
Dr. Holger Ziehr
Pharmaceutical Biotechnology
Telefon +49 531 6181-6001
holger.ziehr@item.fraunhofer.de
Charité – Universitätsmedizin Berlin
Charité’s Medical Department, Division of Infectiology and Pneumonology is intensely dedicated to basic research, besides university-based healthcare and education. The working group headed by Professor Martin Witzenrath is exploring rational foundations for novel therapeutic perspectives and has specialized in translational evaluation of novel therapies. One of its numerous cooperation partners is Leibniz Institute DSMZ. For several years already, the two institutions have collaboratively studied the processes taking place during treatment of bacterial infections with phages.
Prof. Dr. Martin Witzenrath
Medical Department, Division of Infectiology and Pneumonology
Phone +49 30 450553122
martin.witzenrath@charite.de
Charité Research Organisation GmbH
CRO is a wholly-owned subsidiary of Charité – Universitätsmedizin Berlin. The contract research organization is specialized in scientifically demanding and complex phase-I and phase-II trials, which CRO has conducted in its own research unit for over 10 years.
Jeanette Lehmann
Project Manager
Phone +49 30 450539234
jeanette.lehmann@charite-research.org